genetic causes
Periodontal EDS
Both from a scientific point of view and with regard to potential new treatments it is essential to give individuals with EDS a precise clinical and molecular diagnosis with regard to the specific subtypes.
It has been recently shown that pEDS is caused by heterozygous missense or in-frame insertion/deletion mutations in C1R or C1S (13) which encode subunits C1r and C1s of the first component of the classical complement pathway. The classical complement pathway is centrally involved in periodontal host defense, and its over-activation or deregulation may excessively amplify inflammation and contribute to immunopathology. Additionally, the C1q subunit – which binds to the C1s-C1r tretamer – has a triple helical structure which resembles collagen and may explain abnormal interactions with extracellular matrix components (13). Periodontal EDS may thus be due to a combination of excessive complement pathway activation and connective tissue alterations.
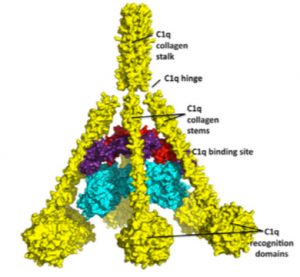
The mentioned C1r and C1s units are part of a C1s-C1r-C1r-C1s tetramer (Figure)which is Ca(2+)-reliant and connected to the recognition protein C1q (49,50). After C1 binding to immune complexes via C1q, C1r is automatically activated and sticks to C1s which itself sticks to C4 and C2 to form the classical pathway C3 convertase (51-53). At first C4 is split into C4a and C4b, and C2 into C2a and C2b. C4b then binds to the antigen and forms a complex with C2a to produce the active enzyme C3 convertase. This C3 convertase digests C3 into C3a and C3b. C5 convertase is formed by the C3b and then activates C5 and initiates the membrane attack complex (MAC). In this MAC C5 is split into C5a and C5b, with the last mentioned to bind to a cell surface and one C6 molecule. Then binding one C7 molecule, which enables the bindig of one C8 molecule. This C8 binds into the cell membrane and allows C9 to bind and polymerize in the cell membrane with a small pore being the result and lysis by osmotic shock is engaged (17).
As part of the periodontal host defense a deregulation as well as an overactivation of the classical complement pathway may intensify inflammation and go hand in hand with immunopathology.